We develop synthesis approaches to fabricate hierarchically structured porous (electro) catalysts based on metallic materials, high-entropy alloys and noble metal oxides. We are particularly interested in H2 generation

Related papers:
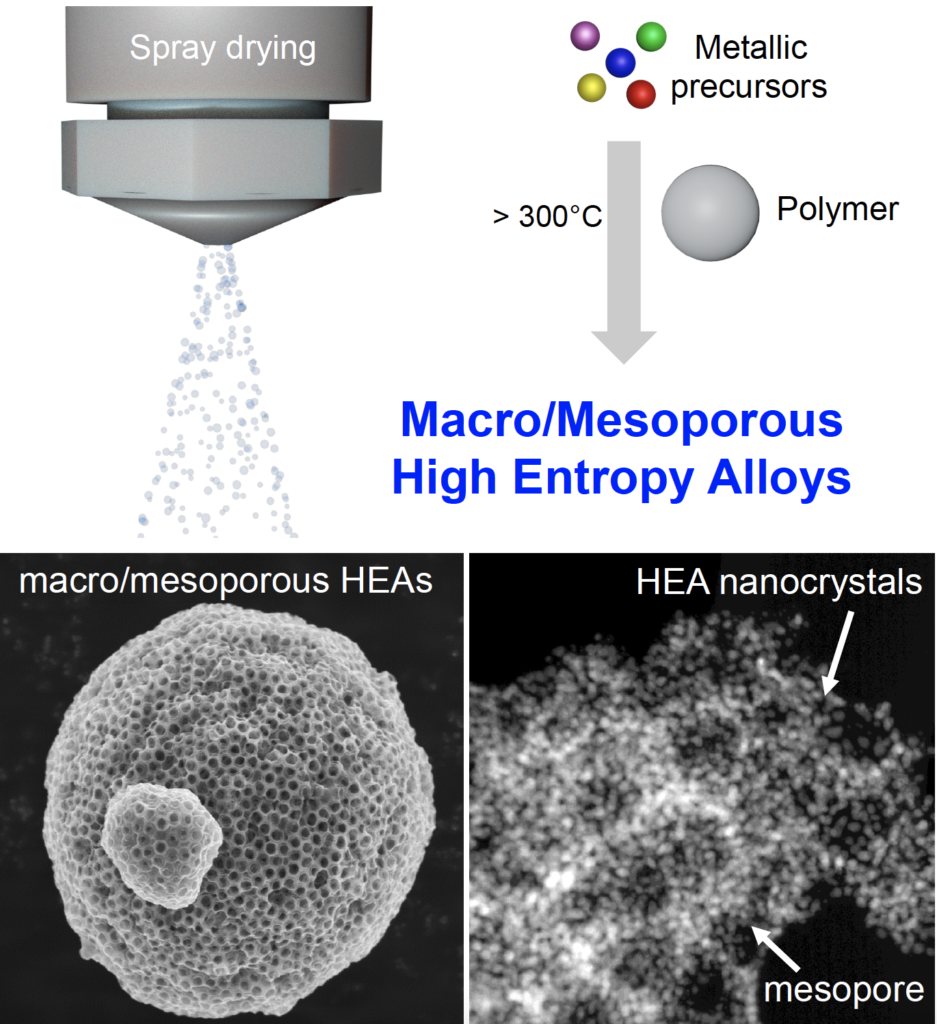
High-Entropy-Alloy Nanocrystal Based Macro- and Mesoporous Materials
M. L. De Marco; W. Baaziz; S. Sharna; F. Devred; C. Poleunis; A. Chevillot-Biraud; S. Nowak; R. Haddad; M. Odziomek; C. Boissière; D. P. Debecker; O. Ersen; J. Peron; M. Faustini, ACS Nano, 16, 15837-15849, 2022.
High-entropy-alloy (HEA) nanoparticles are attractive for several applications in catalysis and energy. However, shaping nano-HEAs into hierarchical structures remains a great challenge. We introduce a simple soft-chemistry route to fabricate ordered macro- and mesoporous materials based on HEA nanoparticles, with high surface area, thermal stability, and catalytic activity toward CO oxidation. The process is based on spray-drying from an aqueous solution containing five different noble metal precursors and polymer latex beads. Importantly, this process is green, scalable, and versatile and could be potentially extended to other classes of HEA materials.
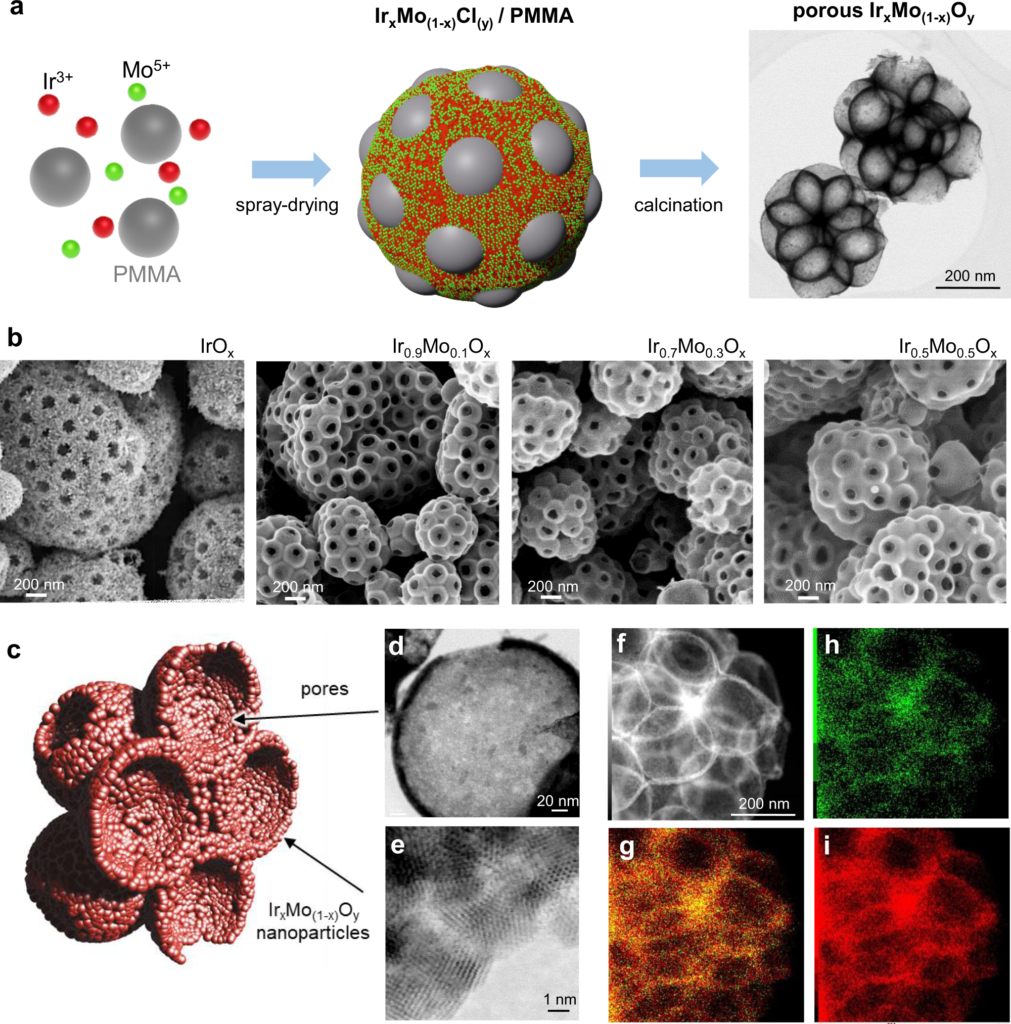
Hierarchically Structured Ultraporous Iridium-Based Materials: A Novel Catalyst Architecture for Proton Exchange Membrane Water Electrolyzers
M. Faustini; M. Giraud; D. Jones; J. Rozière; M. Dupont; T. R. Porter; S. Nowak; M. Bahri; O. Ersen; C. Sanchez; C. Boissière; C. Tard; J. Peron, Advanced Energy Materials, 9, 4, 1802136, 2019
Iridium oxide is the gold-standard catalyst for the oxygen evolution reaction (OER) in acidic media due to its unmatched activity and stability. Here, a new catalyst architecture comprising a nanoneedle network of iridium-containing oxides assembled into macroporous micrometric particles with ≈75% of porosity is reported. The rationally designed porous hierarchical structure optimizes the accessibility of reactants and products to the surface of the nanoparticles and maximizes catalyst activity. The materials are easily prepared from aqueous solutions by an industrially viable spray-drying route through an evaporation self-assembly mechanism.

The origin of the high electrochemical activity of pseudo-amorphous iridium oxides
M. Elmaalouf; M. Odziomek; S. Duran; M. Gayrard; M. Bahri; C. Tard; A. Zitolo; B. Lassalle-Kaiser; J.-Y. Piquemal; O. Ersen; C. Boissière; C. Sanchez; M. Giraud; M. Faustini, J. Peron, Nature Communications, 12, 3935, 2021.
The reasons for the higher electroactivity of amorphous iridium oxides compared to their crystalline counterpart is still the matter of an intense debate in the literature. By producing iridium-based mixed oxides using aerosol, we are able to decouple the electronic processes from the structural transformation, i.e. Ir oxidation from IrO2 crystallization, occurring upon calcination. Full characterization using in situ and ex situ X-ray absorption spectroscopy, X-ray photoelectron spectroscopy, X-ray diffraction and transmission electron microscopy allows to unambiguously attribute their high electrochemical activity to structural features and rules out the iridium oxidation state as a critical parameter.
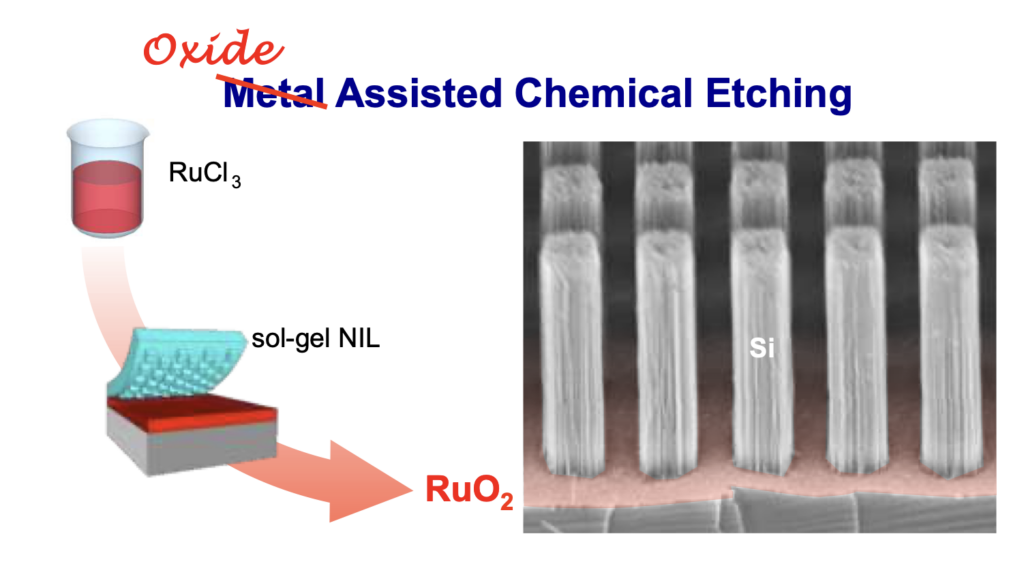
Replacing Metals with Oxides in Metal-Assisted Chemical Etching Enables Direct Fabrication of Silicon Nanowires by Solution Processing
M. Gayrard; J. Voronkoff; C. Boissière; D. Montero; L. Rozes; A. Cattoni; J. Peron; M. Faustini, Nano Letters 21, 5, 2310-2317, 2021
Metal-assisted chemical etching (MACE) has emerged as an effective method to fabricate high aspect ratio nanostructures. This method requires a catalytic mask that is generally composed of a metal. Here, we challenge the general view that the catalyst needs to be a metal by introducing oxide-assisted chemical etching (OACE). We perform etching with metal oxides that can be obtained by direct nanoimprint lithography or block-copolymer lithography from chemical solution on a large scale.